18 Which Best Describes the Bonding in Cu S
Northrups Chem 112 Section TTU General Chemistry. Up to 24 cash back B.

Possible Multiple Choice Questions
Metals with a body-centered cubic lattice contain a net of four metal atoms per unit cell.

. A The forming of the H-Cl bond releases energy. Net dipole but different atoms involved in the bond d molecular formula but a different overall charge. A Mass is the same as weight.
Electrons are shared and the bonding is ionic. A A silicon atom transfers an electron to each oxygen atom. Strong single covalent bonds with weak intermolecular forces.
N-F A I only B II only C III only D I and III E I II and III 3 Which of the following BEST describes the bonding found within solid Al 2 O 3. Apply scientific notations if necessary. Forces among Br2 molecules are stronger than those among Cl2 molecules.
A 50 V 3 b 26 Cr 2 c. Of body centered cube. Select e Use electronegativity values and determine if the O-F bond is classified as covalent polar covalent or ionic.
How many bonding electrons are there in the urea molecule. 32 Explain in terms of electronegativity why an H-F bond is expected to be more polar than an H-I bond. Liquids Solids Practice Quiz 2.
D Mass depends on gravity. Select f Is the OF2 molecule polar or nonpolar. By 2008 however global methane levels which had stayed mostly flat since 1998 had risen to 1800 nmolmol.
Lower because London dispersion forces among C2h5OH molecules are greater than those among CH3OH molecules. The greatest degree of ionic character would be found in a bond between sulfur and A. Which one of the following describes the major intermolecular force in I2 s.
Electrons are shared and the bonding is covalent. Which of the following best describes the type of bonding in. Electrons are transferred and the bonding is ionic.
Which of the following best describes a bond formed between two unlike atoms. When a reaction occurs between atoms with ground state electron con gurations 1s22s1 and 1s22s22p5 the predominant type of bond formed is A. Quartz SiO2 is a solid with a melting point of 1550C.
Therefore it is useful in cooking equipment. Check all that apply---Copper conducts electricity more readily than gold---Copper reacts quickly with air and water to form a tarnish and it corrodes more easily than gold---Copper conducts heat very well. Up to 24 cash back Ahydrogen bonding Bcovalent bonding Cmetallic bonding Dionic bonding 39Which term represents an intermolecular force in a sample of water.
Each 33 Given the reaction. Which statement best describes ionic bonding. Chem 1120 - Chapter 11.
It is the electrostatic attraction between positive ions and delocalized electrons and occurs by the. Lig Li g e-a electron affinity b binding energy c ionization energy d electronegativity e none of these 5. Raoults Law relates the vapor pressure of the solvent above the solution to its mole fraction in the solution.
What is the coordination no. Which statement is correct. A lattice of positive and negative ions held together by electrostatic forces.
40Base your answer to the following question on Which pair of characteristics describes the molecule illustrated below. 10182019 Chemistry High School answered expert verified What best describes the bonding in a silicon dioxide molecule. The concentration of methane in Earths atmosphere in 1998 expressed as a mole fraction was 1745 nmolmol ppb.
NH 3 because nitrogen forms a single bond with each hydrogen atom. A Strong covalent bonds between atoms with similar electronegativities. Asymmetrical and polar Bsymmetrical and nonpolar Casymmetrical and.
The electrons are in fixed positions between the nuclei of metal atoms. 2 pts BONUS Questions 1 pt. Which statement best describes what is happening in this model.
2 Which of the following bonds would be best categorized as covalent. NH3 17034 gmol CuO 7955 gmol N2 2802 gmol Cu 6355 gmol H20 18016 gmol Please. Which of the following terms accurately describes the energy associated with the process.
Which of these best describes why copper is considered to be a more useful metal than gold. Play this game to review Chemical Bonds. The electrons are moving easily between the nuclei of metal atoms.
The electrons are floating to the top of the nuclei of metal atoms. C Raoults Law works best when applied to dilute solutions. A They contain both a nonpolar group and a polar group.
Perform the following calculation maintaining the correct number of significant figures. The type of bond is determined by. B Mass and weight are always proportional to each other.
Atomospheric methane is a potent greenhouse gas. The electrons are sinking down between the nuclei of metal atoms. Compared to the equilibrium vapor pressure of CH3OH l at 300 K the equilibrium vapor pressure of C2H5OH l at 300 K is.
Which of the following is an accurate statement. 2014-01-27 091800. B A silicon atom receives two electrons from each oxygen atom.
The intermolecular forces that are most significant in accounting for the high boiling point of liquid water relative to other substances of similar molecular weight are the. The species that contains 24 protons 26 neutrons and 22 electrons would be represented by the symbol. A Raoults Law applies exactly to all solutions.
C A silicon atom shares an electron pair with each oxygen atom. The bonding in quartz is best described as. O x 18 g H 2 O 36 g H 2 O produced.
B Raoults Law works best when applied to concentrated solutions. NH 4 because nitrogen needs four electrons and each hydrogen needs one electron. Electrons are transferred and the bonding is covalent.
NH 4 because nitrogen needs one electron and each hydrogen needs four electrons. NH 3 because nitrogen forms a triple bond with each hydrogen atom. Select d Which best describes the F-O-F bond angles in OF2.
H2 Cl 2 2HCl Which statement best describes the energy change as bonds are formed and broken in this reaction. 0 g C 2 H 2 40 g O 2 176 g CO 2 36 g H 2 O. Strong multiple covalent bonds including pi bonds with weak intermolecular forces.
2 mol C 2 H 2 mole Final answers. Which of the following best describes the type of bonding in a sample of CO2 s answer choices. Which statement best describes the chemistry of soaps.
C Weight depends on gravity.

Amino Acids Polypeptides And Proteins Ppt Download

Quiz Periodic Trends Jennifer Lopez Worksheet Periodic Trends 1 Which Statement Best Describes Group 2 Elements As They Are Considered In Order From Course Hero

Family Like Branches On A Tree We All Grow In Different Directions Yet Our Roots Remain As One Family Quotes Inspirational Quotes Peace Quotes

Beautiful Words To Describe My Daughter Daughter Words Matter Words

Let S Explore This Suggested Time Allotment 15 Minutes Rate The Statements That Best Describes You By Brainly Ph

Atomic Structure Periodic Table Science Quiz Quizizz

Chem 180 Set 2 Flashcards Quizlet

Ccr Biology Chapter 4 Practice Quizzes Summer

The H O H Bond Angles In H3o Are Approximately 107 The Orbitals Used By Oxygen In These Bonds Are Best Described As

Chapter 11 Modern Atomic Theory Ppt Video Online Download

Electronic Structure Of Atoms Ppt Download
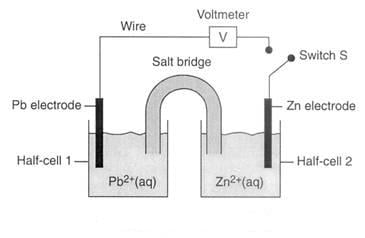
The Electrochemical Series Proprofs Quiz
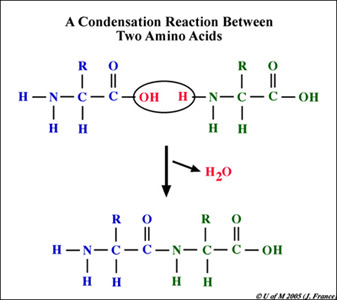
Khan Academy Introduction To Biological Molecules Flashcards Quizlet



Comments
Post a Comment